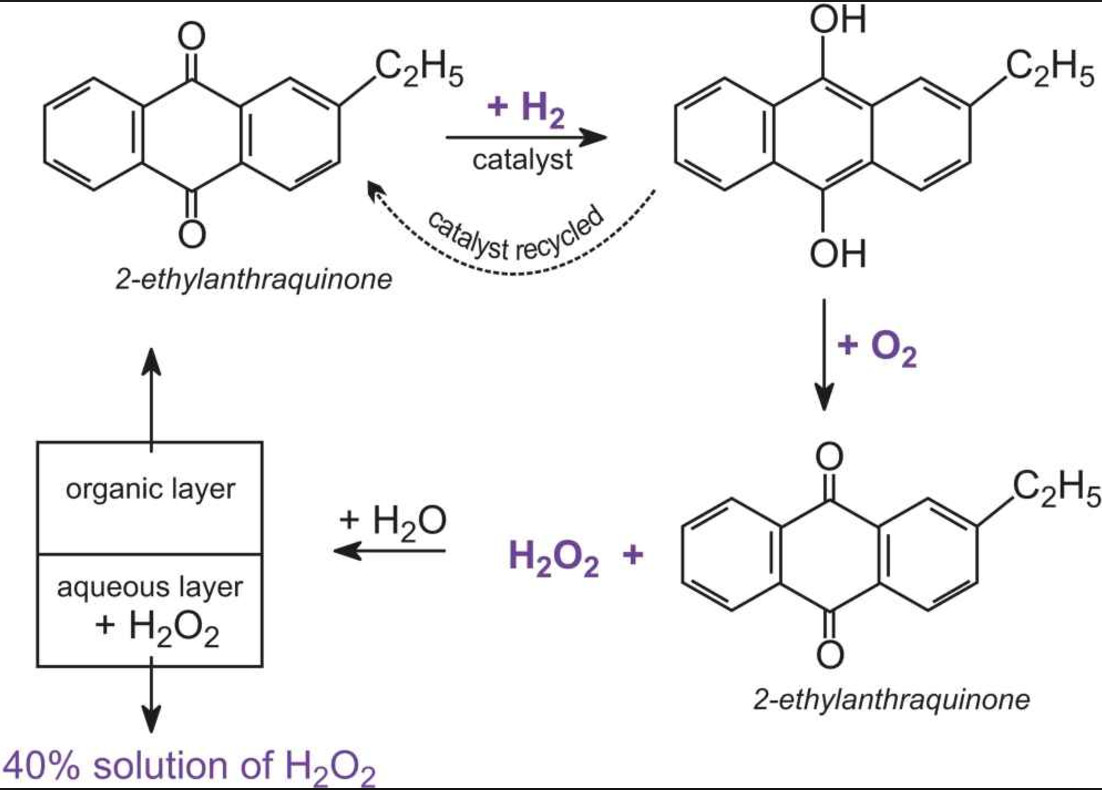
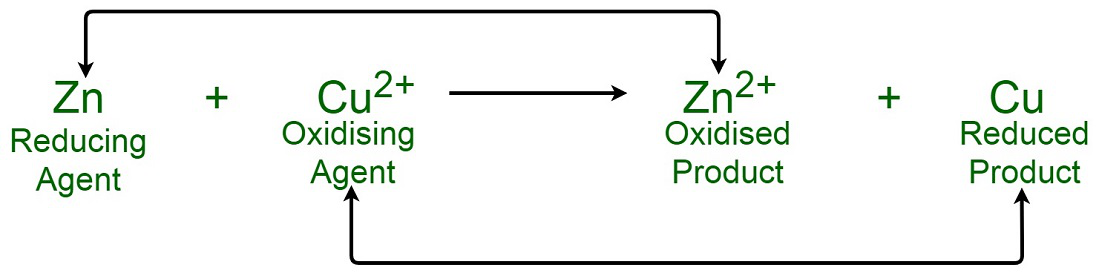
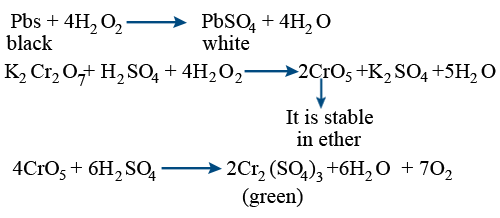
While sulphur dioxide and hydrogen peroxide can act as oxidising as well as reducing agents in their reactions, ozone and nitric acid act only as oxidants. Why ?

Hydrogen peroxide acts both as an oxidising and as a reducing agent depending upon the nature of the reacting species. In which of the following cases H(2)O(2) acts as a reducing agent

Hydrogen peroxide can act as either an oxidizing agent or a reducing agent, depending on the species present in solution. Write balanced half-reaction equations for each of the following: (a) H2O2(aq)