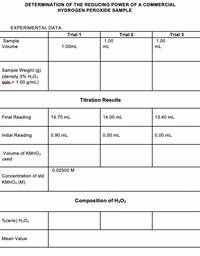
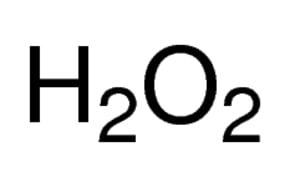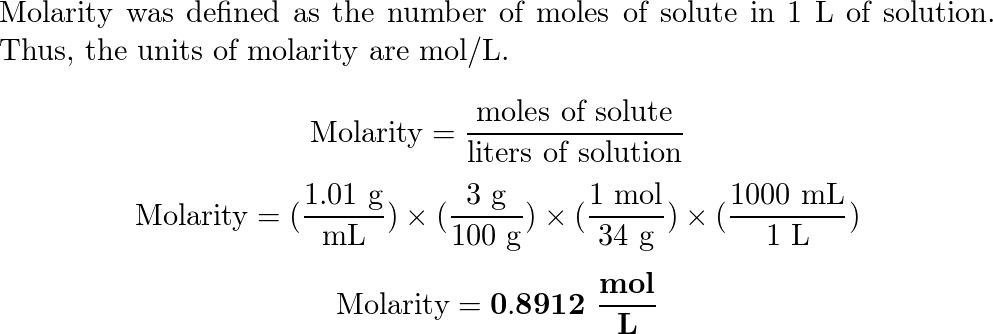OneClass: If I have hydrogen peroxide, molecular weight(g/mol) is 34.02, its density (g/ml) is 1.2, i...

I have H2O2 of molecular wt 34.01gm and 30% w/v. What does it mean that I am not getting it and I want to prepare 0.1M solution, how can i? | ResearchGate

Table 1 from Biodecomposition of Hydrogen Peroxide (H 2 O 2 ) in Water and in Organic Solvents Using Saccharomyces cerevisiae Meyen ex E.C. Hansen (Fungi: Ascomycota) | Semantic Scholar

SOLVED: You are given an aqueous solution of hydrogen peroxide that is 30.0% by mass and has a density of 1.11 g/mL What is the molality of the solution? Molar mass of

The strengths of 5.6 volume hydrogen peroxide (of density 1g/ mL) in terms of mass percentage and molarity (M) , respectively are : (Take molar mass of hydrogen peroxide as 34g / mol )

gchem2 - quiz 1 - 1. A solution of hydrogen peroxide is 30.0% H2O2 by mass and has a density of 1.11 g/cm . The molarity of the solution is: (hint: | Course Hero