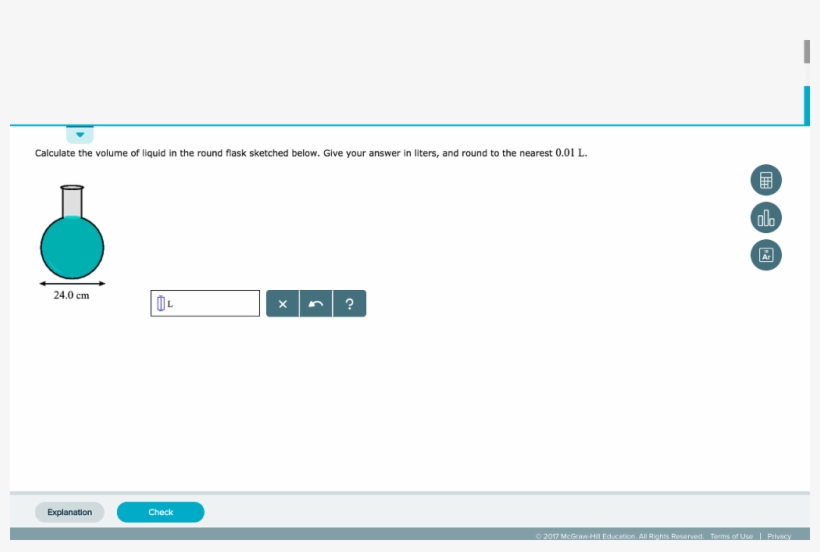
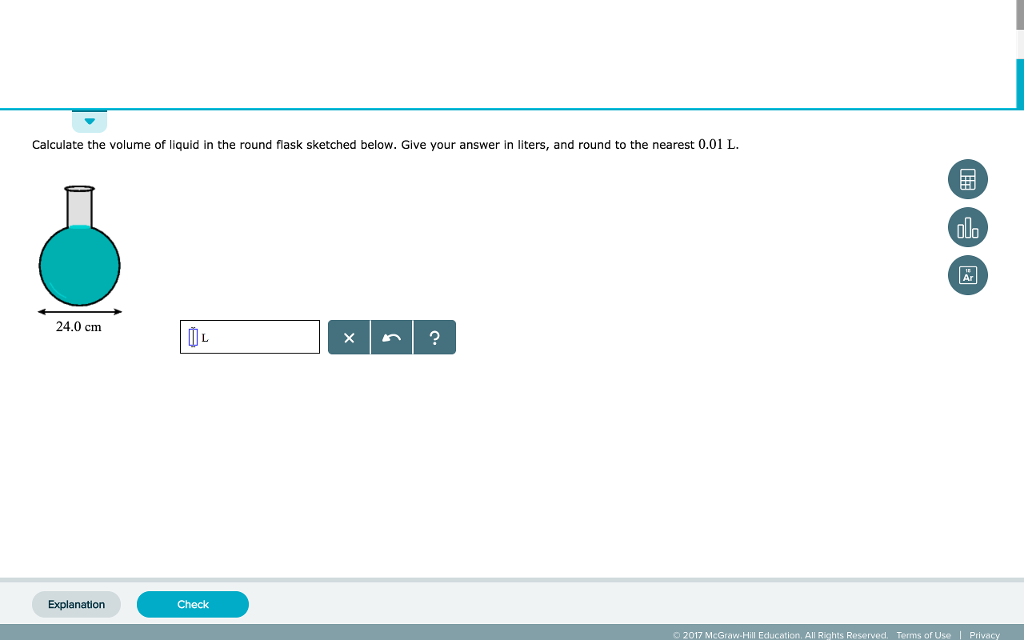
SOLVED:To find the volume of a flask, the flask is evacuated so it contains no gas. Next, 4.4 g CO2 is introduced into the flask. On warming to 27^∘ C, the gas
What is the volume of a conical flask which is 13 cm in height, with base radius of 6 cm, and an upper radius of 2 cm? - Quora

Consider the flasks in the following diagrams: Assuming the connecting tube has negligible volume, predict what each diagram will look like once the stopcock between the two flasks is opened. Calculate the

On , heating a flask at constant pressure, from 27^o C to 227^o C , 200mL of air was expelled out. what was the volume of the flask?

A glass flask of volume one litre at 0^o C is filled level full of mercury at this temperature. The flask and mercury are now heated to 100^o C. How much mercury

A flask was heated from 27^(@)C to 227^(@)C at constant pressure. Calculate the volume of the flask if 0.1 dm^(3) of air measured at 27^(@)C was expelled from the flask.


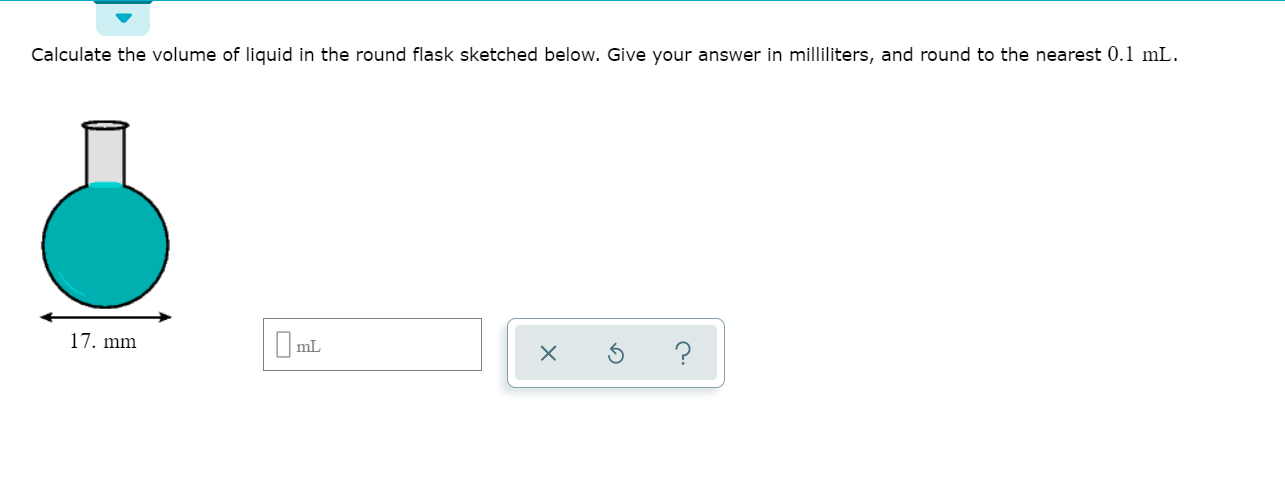

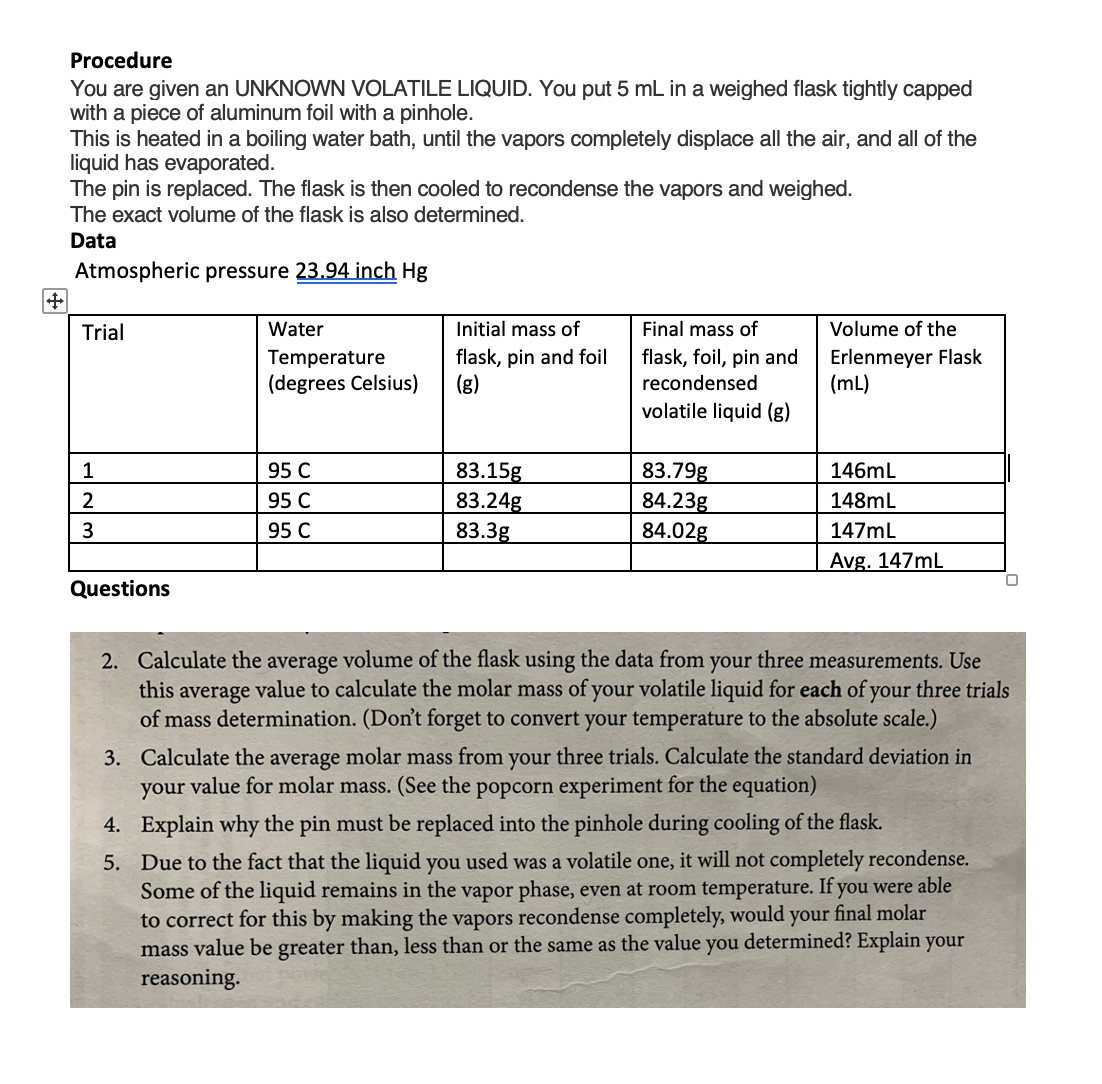
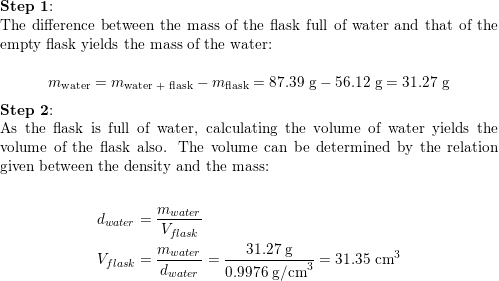

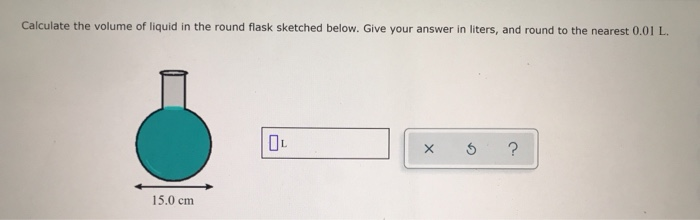
:max_bytes(150000):strip_icc()/GettyImages-493151728-c08e9b2d60bb401b8bbdd2e66de7a93b.jpg)